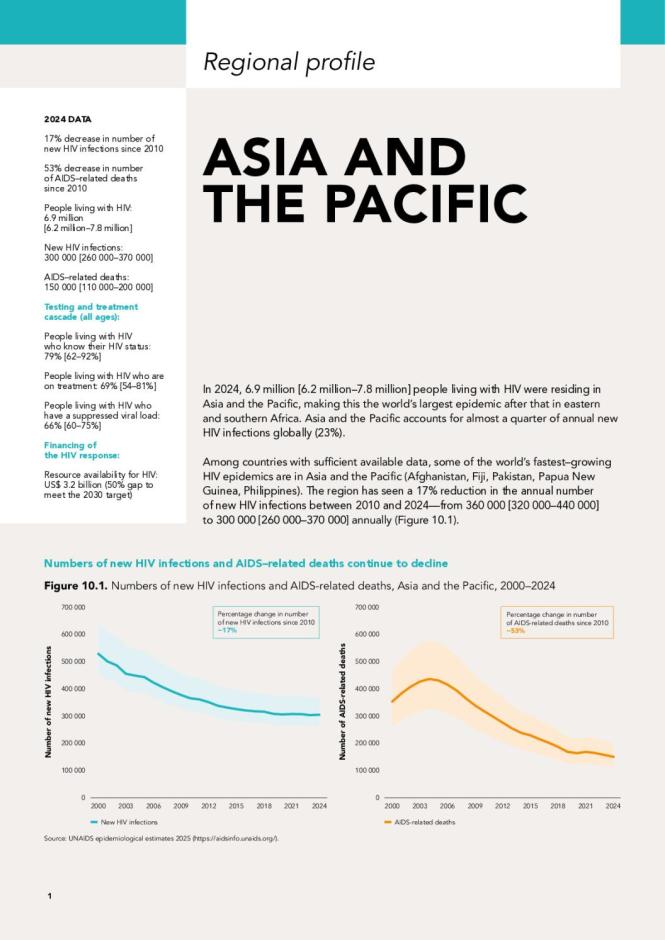
HIV Drug Resistance Surveillance in Countries Scaling Up Pre-exposure Prophylaxis
This technical guidance describes the methods and implementation considerations to monitor the prevalence of HIV drug resistance among PrEP users diagnosed with HIV through a cross-sectional survey. The outcomes of the survey will be used to inform the selection of maximally effective first-line combination antiretroviral therapy for PrEP users who acquire HIV.